
Unlocking the Secrets of Aging: The Vital Role of NAD+
, by Dylan Arrazati, 7 min reading time

, by Dylan Arrazati, 7 min reading time
Unlocking the Secrets of Aging: The Vital Role of NAD+
NAD+: The Cellular Helper
At its core, NAD+ serves as a critical coenzyme, a molecular assistant that enables enzymes to perform essential reactions vital for cellular functions. It's the linchpin in regulating mitochondrial activities and energy metabolism within our cells.
The Aging Challenge
Here's the conundrum: As we age, our cellular NAD+ levels gradually decline. This decline threatens to disrupt metabolic equilibrium, with far-reaching consequences. Reduced NAD+ levels make our genetic blueprints—our DNA—vulnerable to unwarranted changes. Notably, animal studies have strongly suggested that these changes pave the way for numerous age-related health conditions, such as neurodegeneration. In light of these findings, scientists are on a mission to uncover innovative strategies for preserving NAD+ levels.
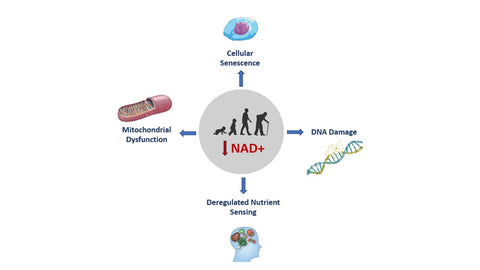
Cellular Consequences of NAD+ Decline
To grasp the significance of NAD+ in aging, we must first explore the cellular fallout of its decline.
Mitochondrial Dysfunction: Mitochondria are the powerhouses of our cells, and they rely heavily on NAD+ for optimal performance. As NAD+ depletes with age, we become more susceptible to mitochondrial dysfunction, which drives oxidative stress and inflammation, two notorious drivers of aging. Thus, finding ways to replenish NAD+ levels is paramount to preserving mitochondrial health. Notably, boosting NAD+ levels also helps stimulate the removal and recycling of defective mitochondria, which studies have shown contributes to longevity and even the prevention of inflammation-driven diseases.
DNA Damage and Cellular Senescence: Aging takes its toll on our DNA, accumulating damage from various sources like solar radiation and inflammation. To counter this genetic wear and tear, our cells employ a group of guardian proteins called sirtuins. These compounds rely on NAD+ for activation, yet dwindling NAD+ levels with age hinder sirtuin activity, exacerbating DNA damage.
(The Consequences of Declining NAD+ Levels)
Promising Effects of Elevated NAD+
Animal studies tantalizingly suggest that increasing NAD+ levels could be a game-changer. These interventions extend lifespan and enhance healthspan—the number of healthy, productive years. Moreover, NAD+’s benefits extend to improved physical function, delayed muscle degeneration, inhibition of bone deterioration, enhanced tumor suppression, and even extended lifespan in experimental mice.
The heart and brain are arguably the two most essential organs for sustaining life, as the heart pumps oxygenated blood to supply essential nutrients to all organs, while the brain coordinates and controls the body's functions. Researchers have found that augmenting NAD+ levels can mitigate cardiac hypertrophy, delay blood vessel aging, and protect against age-related cognitive decline.
Interestingly, manipulating NAD+ metabolism with NAD+ boosters has proven effective in curbing diet and age-related weight gain in rodents. Furthermore, replenishing NAD+ levels has demonstrated benefits in countering obesity and reversing diabetic symptoms, especially in female mice.
Synthesizing Cellular NAD+ Levels
Understanding how our cells synthesize NAD+ is essential in the quest to preserve this vital coenzyme. NAD+ is crafted through three distinct pathways: the de novo pathway, the Preiss-Handler pathway, and the salvage pathway.
De Novo Pathway: This pathway uses amino acids, such as tryptophan, to initiate NAD+ synthesis "from scratch." Tryptophan, sourced from dietary items like meat, fish, and cheese, undergoes a series of chemical reactions within the cell's cytosol to produce NAD+.
Preiss-Handler Pathway: This alternative route utilizes nicotinic acid (NA) found in various foods like meat and nuts. Interestingly, this pathway intersects with the de novo pathway, converging on the production of nicotinic acid mononucleotide (NAMN), a crucial intermediate step on the path to NAD+ production.
Salvage Pathway: This is perhaps the most vital NAD+ synthesis pathway, as it recycles and salvages NAD+ breakdown products to generate new NAD+. Our cells use vitamin B3 variants like nicotinamide (NAM), NA, and nicotinamide ribose (NR) in this intricate process. NAD+ can be dismantled into NAM, which, with the help of an enzyme called NAMPT, can be converted back into NAD+ via its direct precursor, NMN. This elegant recycling process rejuvenates the NAD+ cycle.
Steps to Replenish NAD+
The central question is: How can we replenish declining NAD+ levels? Scientists are exploring a range of strategies, with several promising avenues.
NAD+ Precursors: Among the most potent methods is the use of NAD+ precursors, which circumvent the challenges of directly supplementing NAD+. Two leading precursors, Nicotinamide Mononucleotide (NMN) and Nicotinamide Riboside (NR), have demonstrated the ability to significantly boost circulating NAD+ levels in both rodents and humans. Their benefits extend to improved physical performance, reduced inflammation, and enhanced cognitive function, even in models of Alzheimer's disease.
Activating NAD+ Synthesis and Inhibiting NAD+ Breakdown: Aging disrupts the balance between NAD+ synthesis and consumption. One key contributor to NAD+ depletion is the heightened activity of CD38, an enzyme that breaks down NAD+. Researchers are exploring CD38 inhibitors to restore NAD+ levels and potentially slow the aging process.
Another factor in NAD+ decline is increased DNA repair, which recruits NAD+ through an enzyme called PARP1. Inhibiting PARP1 has been shown to increase NAD+ levels, but researchers must carefully balance the benefits with potential downsides, as limiting PARP1 activity could hinder DNA repair processes.
One promising approach involves resveratrol, a plant-based antioxidant compound known as a polyphenol. Resveratrol can enhance the activity of NMNAT, the enzyme responsible for converting NMN to NAD+. This compound offers an avenue for preserving NAD+ levels while supporting healthy aging.
Final Thoughts:
The quest for effective aging interventions is reaching new heights with the emergence of NAD+ as a potent player. This powerful coenzyme wields considerable influence over our cellular health, impacting mitochondrial function, DNA repair, and cellular senescence. As science continues to unravel its secrets, we find ourselves on the brink of a new era in our quest to age gracefully and healthily. The journey is far from over, but the promise of a healthier, more vibrant future grows stronger with each discovery.